A device is an instrument, apparatus, implant, or other similar article that is used in the prevention, diagnosis, or treatment of illness or disease. It can also be used for detecting, measuring, restoring, correcting, or modifying the structure or function of the human or animal body for a health purposes. Devices do not rely on metabolism or any other chemical action for their primary intended purpose.
Devices are assigned to one of three regulatory classes (I, II, or III) based on the level of control necessary to assure the product’s safety and effectiveness. The risk the device poses to a patient and/or user significantly influences its class determination. Defining the product’s intended use and indications for use is essential to determine whether you have a medical device and for device classification. For more information, visit this page on the FDA site.
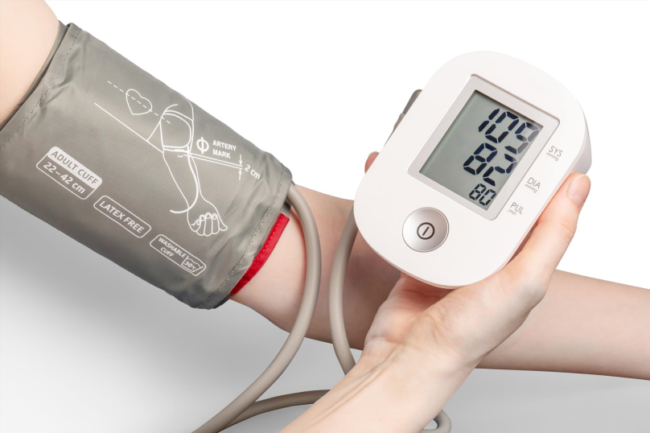
Class II devices are moderate-risk products that must adhere to General Controls. Although they carry greater risk than Class I devices, the risks can be mitigated to an acceptable level by ensuring the product meets certain performance standards or requirements, called Special Controls. Examples of Class II devices are syringes and daily wear contacts meant for daytime use; extended wear contacts are classified as Class III devices. To market a Class II device intended for human use, one must file a premarket notification with the FDA, also known as a 510(k) submission. This submission requires identifying an already marketed predicate product to which the new device must show Substantial Equivalence. Once a Class II device receives FDA clearance, it can be distributed commercially.
Under certain circumstances, a device may not be subject to the above requirements. Specifically, a device may be exempt if it meets the following criteria: it is noninvasive, it does not require an invasive sampling procedure that poses significant risk, it does not introduce energy into a subject by design, and it is not intended for use in diagnostic procedures. For more information on Class II device exemptions visit this page on the FDA site.