SC CTSI Participates in Accrual to Clinical Trials (ACT) Network Launch
The ACT Network helps clinical investigators explore and validate feasibility for clinical studies using electronic health record data. It will be available at USC in the Fall of 2018.
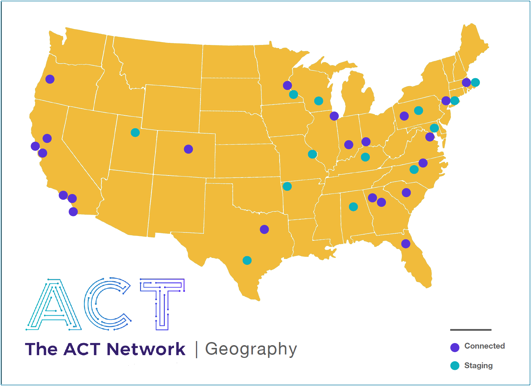
The Accrual to Clinical Trials (ACT) Network was developed collaboratively by members of the CTSA Program consortium to help clinical investigators explore and validate feasibility for clinical studies using electronic health record data. It is an NCATS CTSA Program-funded, real-time, open access platform. This month, it is launching at 21 sites across the CTSA Program consortium. It will be available at USC in the Fall of 2018.
Using ACT, investigators can:
- Explore patient populations in depth, in real time, from the desktop.
- Confirm study feasibility by iteratively testing and refining inclusion and exclusion criteria.
- Demonstrate feasibility in funding proposals and IRB submissions.
- Identify potential partners for multi-site studies by searching for patients across the CTSA network.
"ACT is secure, HIPAA-compliant and IRB-approved, and is available to all researchers at participating sites," says Daniella Meeker, PhD, director of clinical research informatics at the SC CTSI. An additional 13 CTSA’s are now completing technical staging to join ACT later this year, bringing ACT’s reach to nearly 60% of the CTSA Program consortium by the end of 2018.



