SC CTSI Regulatory Science student research team presents Translational Science-focused poster at annual meetings
Student team highlights work on ReguVision™, a tool that leverages drug approval data to visualize key milestones and timelines.
Bringing novel therapies to market can be complicated, and the processes opaque. A team of researchers composed of students and supervised by faculty and staff at the D.K. Kim International Center for Regulatory Science located within the University of Southern California’s Alfred E. Mann School of Pharmacy and Pharmaceutical Sciences are working together to find a scalable solution to address this issue. Faculty and staff members affiliated with the Southern California Clinical and Translational Science Institute (SC CTSI), including from within the Regulatory Knowledge and Support core, serve as mentors and advisors to the student team and projects overall.
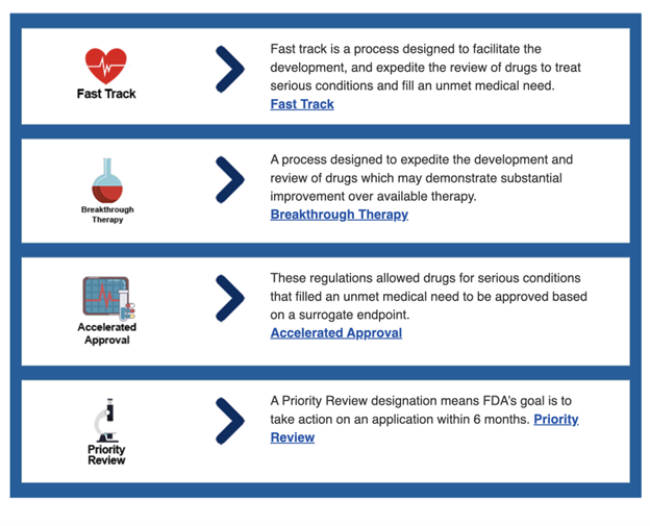
ReguVision™ is a data visualization tool that outlines multiple options and identifies the most efficient path to obtaining approval from the United States Food and Drug Administration (FDA). Four key programs, called Facilitated Regulatory Pathways (FRPs), include Fast Track, Breakthrough Therapy, Accelerated Review, and Priority Review, and are often used in combination to shorten development and review time for serious conditions.
FRPs can significantly speed up the development and approval of treatments for serious or life-threatening conditions, which helps promising therapies reach patients sooner. This represents a significant finding related to Translational Science, which is a priority area for the National Center for Advancing Translational Sciences, a center that funds CTSA hubs around the country, including the SC CTSI. Translational Science drives innovations that address persistent barriers across the translational research pipeline. These advancements span scientific, financial, operational and administrative domains, reshaping how research is conducted to make it faster as well as more efficient and impactful.
Led by the guidance of faculty member Lawrence Liberti, PhD, who is the Director of the Kim Center and an Associate Professor of Regulatory and Quality Sciences at the Mann School, ReguVision™ is funded by a grant from the USC Undergraduate Research Associates Program. The student team is led by Rushanaaz Sokeechand and includes Ana Chi, Daniel He, Jackson Hanf, and Abhiram Gunna. Hanf and Chi represented the team by presenting a poster on this work at the Drug Information Association annual global meeting in 2025. Sokeechand, Gunna, and He presented the poster at the USC Undergraduate Symposium. The poster won second place in the Digital Technology category at the Undergraduate Symposium.
“Working alongside the team has been an enriching experience as a regulatory scientist and a leader,” said undergraduate student collaborator Rushaanaaz Sokeechand. “This project holds great potential to impact approval timelines and showcases the benefits of using FRPs. The data collection process has been challenging, especially with certain submission dates, but the team has been working together to create a detailed database.”
They found that novel drug applications using FRPs achieve faster development and approval timelines on average, with certain pathway combinations offering the greatest efficiency. According to their work, individuals seeking greater time savings can optimize by strategically selecting and combining FRPs, which in turn can lead to more predictable approval timelines and lower the cost of research.
“This project has given the student research group the opportunity to deeply investigate the role of FRPs in the U.S. and to understand ways to optimize the collection and analysis of regulatory information that is often difficult to obtain and interpret,” Liberti said.
The next step is to develop an interactive visualization tool that can illustrate the various approval pathways using an “metro-map” approach, which uses the analogy of a subway map with journeys, connections, and decision points (like stations) to showcase and simplify complex processes.
"This project was so meaningful to me because we took an opaque, complicated process and turned it into a clear, visual strategy,” said undergraduate student collaborator Jackson Hanf. “Developing the metro-map approach allowed us to simplify the FDA approval journey into a navigable path, proving that data visualization can be a powerful tool for lowering the barriers that keep life-saving treatments from the people who need them.”
Grateful to the student team for their contributions, faculty members continue to explore visualization technologies and opportunities to optimize access to the ReguVision™ project by researchers and medicine developers.



