SC CTSI Helps Researchers Navigate Research Bias
Bias can occur in the planning, data collection, analysis, and publication phases of research.
"Let's say you reach in to grab a mouse to run some tests on. Which mouse do you grab? The most active one? The largest one?" This is a question that many inexperienced lab researchers don't ask themselves or even consider. But it is a question that needs to be asked, because it highlights a potential problem in research - Bias.
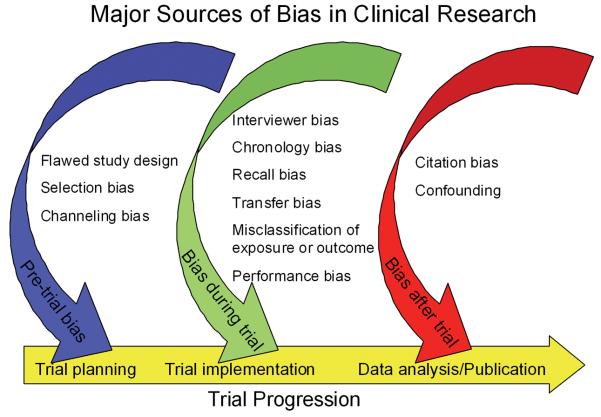
Source: Identifying and Avoiding Bias in Research
"Bias equals error," said Jonathan Samet, MD, MS, Director of the SC CTSI Education, Career Development, and Ethics program and first speaker during the short course on "Bias in Clinical and Translational Research," which was well-attended by faculty and clinicians from USC and CHLA.
Bias can occur in the planning, data collection, analysis, and publication phases of research. A thorough understanding of bias and how it affects study results is essential for the practice of evidence-based medicine.
Types of Bias
Three general types of bias exist: information, selection, and confounding. "Selection bias is what you really have to be wary of in translational research," said Samet. “It occurs when there's an inherent error in the way that individuals or groups are selected to take part in a study. It can lead to complicated and difficult to control bias.”
Information bias occurs from errors in measurement. For example, if a study uses self-reporting data from the study participants, then information bias will most likely be present. "If you ask people how much they weigh, no one will respond by over-reporting their weight," explained Samet.
Confounding bias occurs when the effect of one factor is being studied and another factor becomes mixed up with the factor that is being studied. For example, conducting a smoking study but using people that are overly exposed to asbestos is a good example of confounding bias.
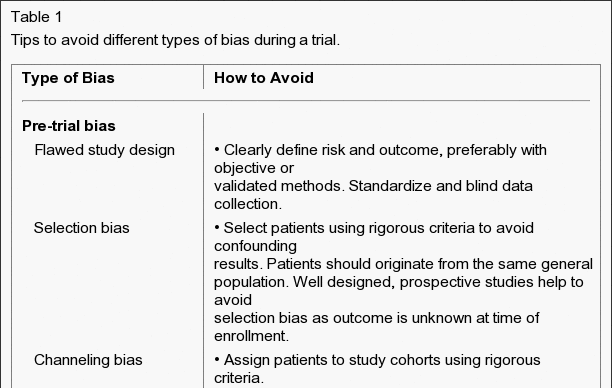
Bias and Data Analysis
The second portion of the course, "Analyzing Data and Interpreting the Results", was presented by Wendy Mack, PhD, Director of the SC CTSI Biostatistics Resources program.
She walked the attendees through a sample study, giving advice along the way about potential bias, alternative study designs, and methods to explore and present the data in well-designed tables and graphs. “A poorly designed graph can actually convey the wrong information,” said Mack.
Throughout an exercise involving a real-life cohort study which analyzed congenital anomalies in multi-ethnic births in the United Kingdom, the attendees then tested their new knowledge and identified potential areas of bias of the study.
Helpful resources to identify and deal with research bias
There are several methods. The best approach to control biases is a well-designed study that has carefully considered and dealt with potential biases. To control certain types of bias (information and confounding bias), a knowledgeable researcher may use statistical models. Researchers can also request a consultation and get expert help from USC faculty and senior staff in this and related fields.
This short course is the 4th of a five-course series sponsored by the SC CTSI. It is meant to provide a broad overview of clinical and translational research.





